Metabolism
on-line - the virtual tutorial room
copyright © 2008 - 2015 David A Bender
Do we need to synthesise fatty acids?
The simple answer is yes.
There are two enzymes involved in fatty acid synthesis (one is a large multi-functional protein) and there is only one case report of a genetic defect; a newborn girl with severe nerve and muscle damage. Experimental deletion of the gene for one of these enzymes in mice (knock-out mice) is lethal - the knockout embryos are undeveloped at embryonic day 7.5 and die by embryonic day 8.5.
The more interesting question to ponder is the extent to which we need to synthesise fatty acids to maintain adipose tissue stores of triacylglycerol when our diet provides 30 - 45% of energy from fat. Incubation of isolated liver cells (hepatocytes) or adipose tissue cells (adipocytes) with [14C]glucose in the presence of insulin shows that label is incorporated into fatty acids (and then triacylglycerol) in both tissues. In the liver (but not in adipose tissue) label from glucose is also incorporated into glycogen. Therefore, it is obvious that fatty acids can indeed be synthesised from glucose. Similar studies show that fatty acids can be synthesised from acetyl CoA, which is formed in mitochondria from pyruvate, the end-product of glycolysis (see the exercise on life threatening acidosis in an alcoholic).
Adipocytes were incubated with [14C]acetyl CoA and insulin in a conventional phosphate-bicarbonate buffer or a phosphate buffer with no bicarbonate. After 30 min the lipids were extracted into chloroform-methanol and radioactivity was measured in both the lipid fraction and also the aqueous fraction (this will be unmetabolised acetyl CoA). The results were as follows (figures show dpm ± standard deviation for 5 x replicate experiments):
unincubated control (bicarbonate buffer) |
bicarbonate buffer |
bicarbonate-free buffer |
|
aqueous phase |
10,105 ± 120 |
1025 ± 75 |
10096 ± 432 |
lipid phase |
32 ± 16 |
9067 ± 135 |
24 ± 15 |
What conclusions can you draw from these results?
It is obvious that bicarbonate (or carbon dioxide) is essential for fatty acid synthesis from acetyl CoA.
In the next set of experiments adipocytes were incubated with [14C]bicarbonate in the buffer and non-radioactive acetyl CoA, again together with insulin. The results were as follows (figures show dpm ± standard deviation for 5 x replicate experiments):
unincubated control |
30 min incubation |
|
| aqueous phase | 10,295 ± 154 |
10,196 ± 162 |
| lipid phase | 29 ± 15 |
32 ± 14 |
What conclusions can you draw from these results?
Although bicarbonate is required for fatty acid synthesis from acetyl CoA, no label from bicarbonate is incorporated into the fatty acids formed. This suggests that there is an intermediate that is carboxylated, but then loses the carbon dioxide again. The overall reaction can be drawn as:
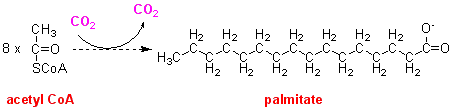
Two enzymes that are required for fatty acid synthesis were isolated from adipocytes. When both together were incubated with [14C]acetyl CoA in a bicarbonate buffer no radioactive palmitate (C16:0) was formed unless NADPH was added to the incubation mixture. Further studies showed that 14 mol of NADPH were oxidised per mol of palmitate formed.
What conclusions can you draw from these results?
Looking at the overview of fatty acid synthesis shown above it is obvious that there must be reduction reactions if acetyl CoA is to form a saturated hydrocarbon chain. Presumably one mol of acetyl CoA forms the carboxyl group of palmitate, but the remaining 7 mol have to be reduced. The stoichiometry (14 mol of NADPH consumed per mol of palmitate formed) suggests that there are two reduction reactions for each acetyl CoA added.
How might you confirm this suggestion?
The obvious experiment would be to incubate the enzymes with acetyl CoA and bicarbonate using [3H]NADPH, and measure the incorporation of label into the palmitate formed.
 Neither
enzyme alone catalysed any synthesis of palmitate or any other fatty acid. Studies
of the smaller enzyme showed that it catalyses the carboxylation of acetyl CoA
to malonyl CoA.
Neither
enzyme alone catalysed any synthesis of palmitate or any other fatty acid. Studies
of the smaller enzyme showed that it catalyses the carboxylation of acetyl CoA
to malonyl CoA.
Further studies of this enzyme showed that it contains the vitamin biotin, bound to a lysine residue at the active site as biocytin, and that the first step in the reaction is formation of carboxybiocytin. This then acts as the donor of the carboxyl group to form malonyl CoA.

This was the first demonstration of a metabolic function of biotin; it is now know to be required for a small number of other carboxylation reactions, including the carboxylation of pyruvate to oxaloacetate in gluconeogenesis (see the exercise on breathless after sprinting).
Having identified malonyl CoA as the product of one enzyme in fatty acid synthesis, studies continued with the second enzyme, incubated with labelled and unlabelled acetyl CoA and / or malonyl CoA. As in previous experiments at the end of a 30 minute incubation lipids were extracted into chloroform-methanol and radioactivity was measured in both the aqueous and lipid phases.
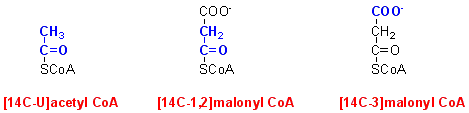
The results were as follows (figures show dpm ± standard deviation for 5 x replicate experiments):
| substrates added | aqueous phrase |
lipid phase |
| [14C-U]acetyl CoA, no added malonyl CoA | 10,095 ± 55 |
26 ± 12 |
| [14C-3]malonyl CoA, no added acetyl CoA | 10,025 ± 30 |
28 ±12 |
| [14C-1,2]malonyl CoA, no added acetyl CoA | 10,132 ± 96 |
32 ± 15 |
| [14C-U]acetyl CoA + non-radioactive malonyl CoA | 8615 ± 64 |
1405 ± 32 |
| [14C-3]malonyl CoA + non-radioactive acetyl CoA | 10,187 ± 55 |
29 ± 13 |
| [14C-1,2]malonyl CoA + non-radioactive acetyl CoA | 267 ± 30 |
9805 ± 67 |
What conclusions can you draw from these results?
The incubations with [14C-3]malonyl CoA confirm the earlier result that although bicarbonate is required to form malonyl CoA, the carbon that was added (carbon-3 of malonyl CoA) is lost again and is not incorporated into the palmitate formed.
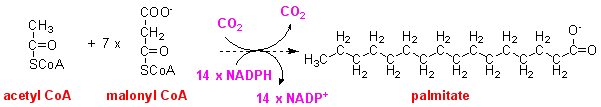
Enzyme 2 obviously requires both acetyl CoA and malonyl CoA to synthesise palmitate. However, only about one seventh as much acetyl CoA as malonyl CoA is incorporated into palmitate. This means that we can draw up the overall reaction of fatty acid synthesis as:
The palmitate formed in the incubations with [14C-U]acetyl CoA + non-radioactive malonyl CoA and [14C-1,2]malonyl CoA + non-radioactive acetyl CoA was subjected to chemical degradation to identify which carbon atoms were labelled in each set of incubations. The results were as shown below.
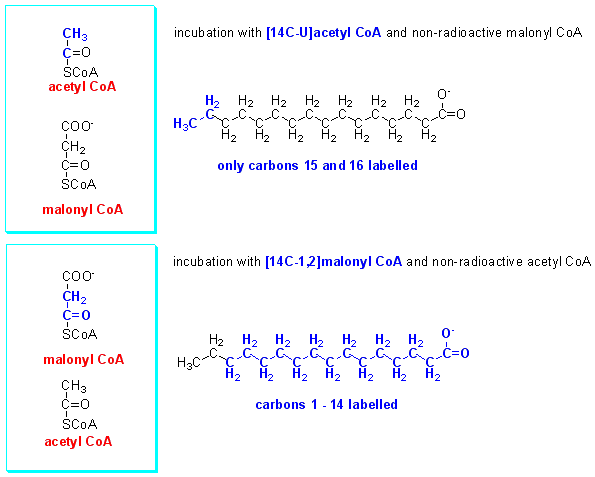
What conclusions can you draw from these results?
It seems likely that carbons 14 and 15 of palmitate arise from acetyl CoA, while the other carbons arise from malonyl CoA, but lose the carbon that was added from bicarbonate during the reaction.
So far we have two enzymes required for fatty acid synthesis. One (acetyl CoA carboxylase) catalyses the carboxylation of acetyl CoA to malonyl CoA, and the other (fatty acid synthase) catalyses the reaction of acetyl CoA plus 7 x malonyl CoA to yield palmitate. No intermediate products between acetyl CoA + malonyl CoA and the final product palmitate can be detected when the mammalian enzyme (or that from yeast) is incubated with acetyl CoA, malonyl CoA and NADPH.

What does this suggest about the mammalian (and yeast) fatty acid synthase?
If none of the intermediate products can be isolated, this suggests that fatty acid synthase is either a multi-enzyme complex, or a single large protein with multiple active sites, and that intermediates are chanelled directly from one active site to the next, until the final product, palmitate, is released.
 In bacteria
and plants a number of intermediates of fatty acid synthesis can be isolated,
and separate enzymes catalysing each step of the pathway can be identified.
The steps involved in fatty acid synthesis were therefore investigated mainly
using the avocado, since this fruit synthesises and contains a considerable
amount of triacylglycerol.
In bacteria
and plants a number of intermediates of fatty acid synthesis can be isolated,
and separate enzymes catalysing each step of the pathway can be identified.
The steps involved in fatty acid synthesis were therefore investigated mainly
using the avocado, since this fruit synthesises and contains a considerable
amount of triacylglycerol.
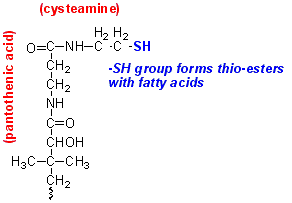 The first
observation was that although the starting substrates are acetyl CoA and malonyl
CoA, both the acetyl and malonyl moieties are transferred onto acyl carrier
proteins (ACP). These are flexible proteins that have a prosthetic group derived
from the vitamin pantothenic acid and the amine cysteamine, derived from the
amino acid cysteine.
The first
observation was that although the starting substrates are acetyl CoA and malonyl
CoA, both the acetyl and malonyl moieties are transferred onto acyl carrier
proteins (ACP). These are flexible proteins that have a prosthetic group derived
from the vitamin pantothenic acid and the amine cysteamine, derived from the
amino acid cysteine.
Like CoA, this prosthetic group forms a thio-ester with fatty acids.
The first reaction of fatty acid synthesis is condensation between the malonyl moiety of malonyl ACP and the acetyl moiety of acetyl ACP, with loss of carbon dioxide:
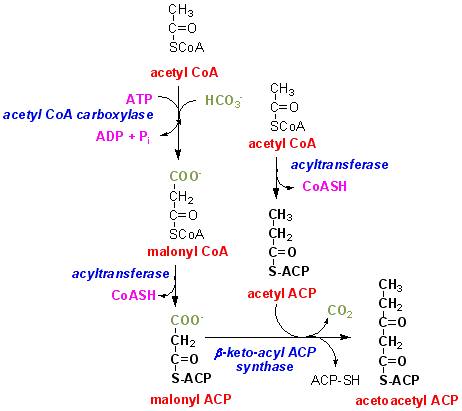
What is the likely next reaction?
We know that there are 14 reduction reactions in the synthesis of palmitate from acetyl CoA, and this suggests that there are two reduction reactions for each acetyl moiety added from malonyl CoA. Therefore, the most likely next reaction is reduction of the oxo-group of acetoacetyl CoA to a hydroxyl group.

How could you test this hypothesis?
Incubation of acetoacetyl ACP with [3H]NADPH will show incorporation of the [3H] label into hydroxybutyrate.
Incubations using [18O]malonyl CoA showed the release of [18O]labelled water. Incubations using [3H]NADPH showed the release of [3H]water.
What do you think is the likely source of this labelled water?
 An
obvious source of water would be dehydration, removing the hydroxyl group on
carbon 3 formed by reduction in the previous reaction and hydrogen from carbon
2, forming a carbon-carbon double bond:
An
obvious source of water would be dehydration, removing the hydroxyl group on
carbon 3 formed by reduction in the previous reaction and hydrogen from carbon
2, forming a carbon-carbon double bond:
What is the likely next reaction?
We know there are two reduction reactions involved, so it is likely that the next reaction is reduction of the carbon-carbon double bond to yield a saturated fatty acyl ACP - in this case butyryl ACP.

We have now synthesised a 4-carbon saturated fatty acid esterified to acyl carrier protein. This can now condense with another molecule of malonyl CoA and undergo the same sequence of reactions. This continues until the product is palmitoyl ACP, when the palmitoyl moiety is cleaved off to form palmitoyl CoA, leaving the acyl carrier protein free to bind acetyl group to start over again.